 Further studies showed that the Bohr model wasnt as accurate as it could be. 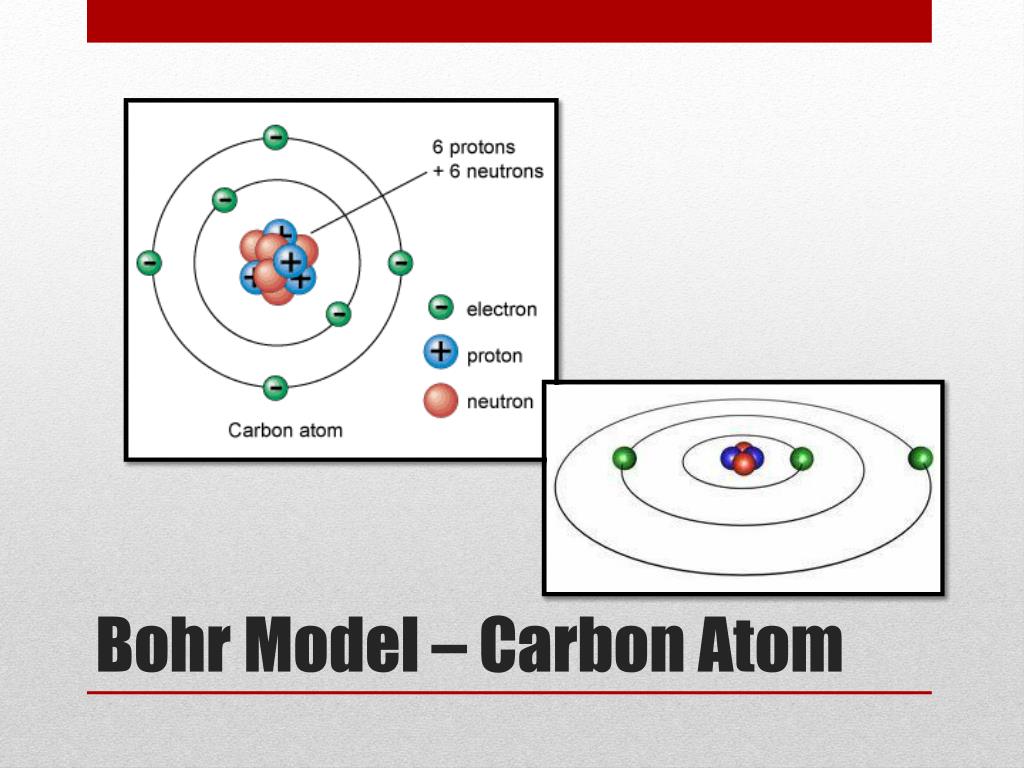
Two of the electrons are in the first energy level while the other five are in the second energy level. However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner. In the Bohr model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. Bohr Model - Includes the element symbol and the electrons in their. For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Thus, the Bohr model for heavier atoms described electron shells. Once the level was full, additional electrons would be bumped up to the next level. Bohr believed each electron orbit could only hold a set number of electrons. More electrons were required to cancel out the positive charge of all of these protons. Heavier atoms contain more protons in the nucleus than the hydrogen atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
